
(Drugs other than Those Specified in Schedules C and C(1), Excluding Drugs in Schedule X)
Introduction
Form 25B is the licence issued under the Drugs and Cosmetics Rules, 1945, for the repacking of drugs intended for sale or distribution in India. It applies specifically to drugs other than those specified in Schedules C and C(1), and excluding Schedule X drugs.
This licence authorises the licensee to obtain bulk drugs or medicines in original sealed containers from duly licensed manufacturers or importers and repack them into smaller pack sizes for commercial distribution, without undertaking any manufacturing activity that affects the composition, quality, or therapeutic value of the drug.
Applicants must fulfil the requirements relating to premises, storage arrangements, technical staff qualifications, equipment, and compliance with Good Manufacturing Practices (GMP) as prescribed in the Rules. Operations under this licence are subject to continuous regulatory oversight, including inspections and adherence to conditions specified in the licence and the provisions of the Drugs and Cosmetics Act and Rules.
Application for licence to manufacture drugs other than those specified in Schedules C and C (1) to the Drugs and Cosmetics Rules (Rule 69)
(1) Application for grant of licence to manufacture for sale or for distribution of drugs, other than those specified in Schedules C and C(l) shall be made to the licensing authority appointed by the State Government for the purpose of this Part (hereinafter in this Part referred to as the licensing authority) and shall be made—
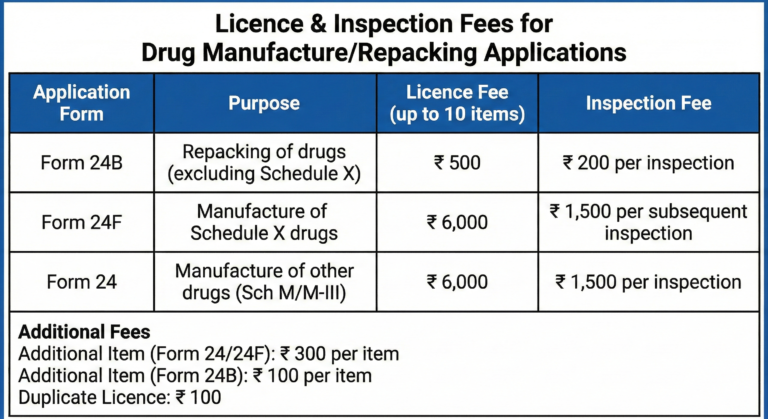
(a) in the case of repacking of drugs excluding those specified in Schedule X for sale or distribution in Form 24B;
(b) in the case of manufacture of drugs included in Schedule X in Form 24F;
(c) in any other case, in Form 24.
(2) (a) Every application in Form 24B shall be made up to ten items for each category of drugs categorised in Schedule M and shall be accompanied by a licence fee of rupees five hundred plus and an inspection fee of rupees two hundred for every inspection
(b) Every application in Form 24F shall be made up to ten items for each category of drugs categorised in Schedule M and shall be accompanied by a licence fee of rupees six thousand and an inspection fee of rupees one thousand and five hundred for every subsequent inspection
(c) Every application in Form 24 shall be made upto ten items for each category of drugs referred to in Schedule M relating to Pharmaceuticals products and Schedule M-III relating to medical devices and in-vitro diagnostics and shall be accompanied by a licence fee of rupees six thousand and an inspection fee of rupees one thousand and five hundred for every inspection.
(4) A fee of rupees one hundred shall be paid for a duplicate copy of the licence issued under clause (a), clause (b) or clause (c) of sub-rule (1) if the original is defaced, damaged or lost.
(5) Applications for manufacture of more than ten items of each category of drugs as referred to in Schedule M relating to pharmaceuticals products and Schedule M-III relating to medical devices and in-vitro diagnostics or for manufacture of additional items of drugs by licensees in Form 24 or Form 24F shall be accompanied by an additional fee at the rate of rupees three hundred for each additional item of drug. Applications in Form 24B for licence to manufacture for sale and distribution for repacking for more than 10 items of each category or for manufacture of additional item of drug shall be accompanied by additional fee of rupees one hundred for each additional item of drugs as categorized in Schedule M and M-III.
(6) Where an application under this rule is for the manufacture of drug formulations falling under the purview of new drug as defined in rule 122E, such application shall also be accompanied with approval, in writing, in favour of the applicant, from the licensing authority as defined in clause (b) of rule 21
Pre-Conditions for the grant of a licence in Form 25B (Rule 71A)
Before a licence in Form 25B is granted the following conditions shall be complied with by the applicant—
(1) the repacking operation shall be carried out under hygienic conditions under the supervision of a competent person;
(2) the factory premises shall comply with the conditions prescribed in Schedule M; and
(3) the applicant shall have adequate arrangements in his own premises for carrying out tests for the strength, quality and purity of the drugs at a testing unit which shall be separate from the repacking unit;
(4) The application for grant of licence for a drug formulation containing single active ingredient shall be made only in proper name:
(5) In case the applicant intends to market the drug under a brand name or trade name, the applicant shall furnish an undertaking in Form 51 to the licensing authority to the effect that to the best of his knowledge based on search in trademarks registry, central data base for brand name or trade name of drugs maintained by Central Drugs Standard Control Organisation, literature and reference books on details of drug formulations in India, and internet, such or similar brand name or trade name is not already in existence with respect to any drug in the country and the proposed brand name or trade name shall not lead to any confusion or deception in the market:
Provided that the repacking units, which, before the commencement of the Drugs and Cosmetics (Second Amendment) Rules, 1977, were making arrangement with institutions approved by the licensing authority for such tests to be carried out on their behalf, may continue such arrangement up to the 31st July, 1977:
Provided further that for tests requiring sophisticated instrumentation techniques or biological or microbiological methods the licensing authority may permit such test to be conducted by institutions approved by it under Part XV(A) of these rules for this purpose.
Explanation—A person who satisfies the following minimum qualifications shall be deemed to be a “competent person” for the purposes of rule 71A or 74A of these rules, namely:—
Post-Conditions for licence in Form 25B (Rule 74A)
A licence in Form 25B shall be subject to conditions stated therein and to the following conditions:—
Duration of licence (Rule 72)
(1) A licence issued in Form 25, Form 25B and Form 25F shall remain valid if the licencee deposits a licence retention fee referred to in sub-rule (2) before the expiry of a period of every succeeding five years from the date of its issue, unless, it is suspended or cancelled by the licensing authority.
(2) The licence retention fee referred to in sub-rule (1) shall be equivalent to the respective fee required for the grant of such licence excluding inspection fee paid for grant of licence.
(3) If the licencee fails to pay licence retention fee on or before the due date as referred to in sub-rule (1), he shall be liable to pay licence retention fee along with a late fee calculated at the rate of two per cent, of the licence fee for every month or part thereof up to six months, and in the event of non-payment of such fee, the licence shall be deemed to have been cancelled.

Disclaimer: The information contained in this Article is intended solely for personal non-commercial use of the user who accepts full responsibility of its use. The information in the article is general in nature and should not be considered to be legal, tax, accounting, consulting or any other professional advice. We make no representation or warranty of any kind, express or implied regarding the accuracy, adequacy, reliability or completeness of any information on our page/article.