Background
The provisions relate to the regulation of prices of scheduled and non-scheduled drug formulations under the Drugs (Prices Control) Order, 2013, which governs price fixation, revision, and monitoring of pharmaceutical products in India. The framework establishes mechanisms for ceiling price determination, retail price fixation of new drugs, permissible margins, and compliance obligations such as price disclosure, reporting, and record maintenance. It also provides transitional provisions from the earlier Drugs (Prices Control) Order, 1995 and prescribes conditions to prevent overcharging, unreasonable price increases, and refusal to sell drugs.
Applicability
The provisions applies to all manufacturers, importers, distributors, and retailers dealing with scheduled and non-scheduled drug formulations regulated under the Drugs (Prices Control) Order, 2013, including formulations listed under the National List of Essential Medicines. The Order does not apply to cases specified as such in Para 32 of DPCO 2013
Compliances under the Order are as follows:
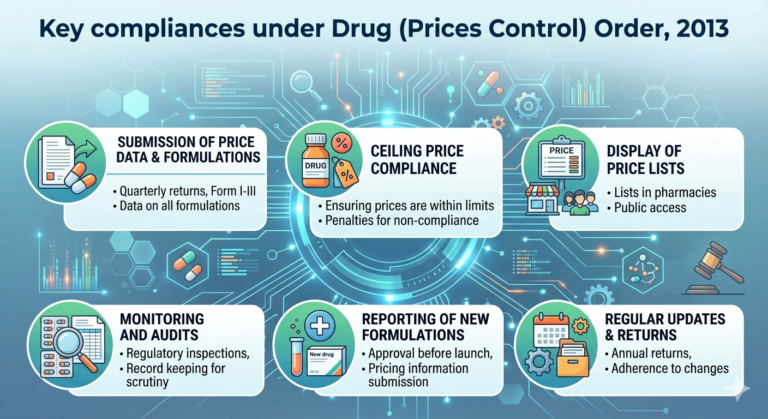
The ceiling price of a scheduled formulation of specified strengths and dosages as specified under the first schedule shall be calculated as per paragraph 4 of Drug (Price Control) Order, 2013.The ceiling price calculated as per sub-paragraph (1) and notified by the Government shall be applicable to scheduled imported formulations also.
Retail price of a new drug available in the domestic market shall be fixed as per paragraph 4(1); however, for a new drug containing molecules or ingredients that are off-patent or nearing patent expiry, the price shall be fixed under paragraph 5(3).
i) price to retailer of a new drug, not available in domestic market, shall be fixed by Govt on principles of “Pharmacoeconomics” of new drug, on recommendation of a Standing Committee of Experts formed under paragraph 15
ii) retail price of such new drug shall be fixed by adding 16% margin to retailer on price to retailer as fixed in item(i)
For a new drug, retail price is determined by reducing 50% of price calculated under paragraph 4(1). If drug is not available in domestic market, its retail price shall be fixed as per sub-para (2). After 1 year from date of such price fixation or from date when “price to retailer” of at least one company is recorded in pharmaceutical market database (whichever is later), retail price for subsequent manufacturers shall be determined as per para4(1). While doing so, prices to retailer of brands belonging to patent holder or those permitted by patentee shall be excluded.
Applies where average price to retailer under para 4(1) causes no reduction in the scheduled formulation price.
a) No reduction in average price to retailer of scheduled formulation
b) Fewer than 5 manufacturers have 1% or more mkt share; ceiling price shall be calculated under Para 6 of Order, 1995
Notwithstanding anything contained in this paragraph, where the price has been fixed and notified by the Government under the Drugs (Prices Control) Order, 1995 the provisions of sub-paragraph (1) shall not apply.
While fixing a ceiling price of scheduled formulations and retail prices of new drugs, 16% of price to retailer as a margin to retailer shall be allowed.
The maximum retail price of scheduled formulations shall be fixed by the manufacturers on the basis of ceiling price notified by the Government plus local taxes wherever applicable, as under: Maximum Retail Price = Ceiling price + Local Taxes as applicable
(1) The source of market based data shall be the data available with the pharmaceutical market data specializing company as decided by the Government and if the Government deems it necessary, it may validate such data by appropriate survey or evaluation.
(2) The Government may prescribe any other appropriate mechanism for collecting or obtaining drug market-based data, and its decision on such data source shall be final.
(3) For first-time ceiling price fixation of scheduled formulations under this Order, market-based data of May 2012 shall apply.
(4) For retail price fixation of new drugs available in the market, data for the month ending immediately before six months from receipt of the pricing application shall apply.
(5) For formulations covered under DPCO, 1995, the reference date shall be as specified in paragraph 10 of this Order.
(6) The Government may, where necessary, use market-based data of any month for fixing or revising ceiling prices.
Prices of scheduled formulations also listed in Ist Sch. to Drugs (Prices Control) Order, 1995 & notified up to 31 May 2012 remained valid up to 30 May 2013. Manufacturers could revise prices based on annual wholesale price index, following which ceiling price was to be fixed under para 4(1).Prices of scheduled formulations covered under DPCO, 1995 but not included in the First Schedule of this Order, and notified up to 31 May 2012, shall remain effective up to 30 May 2013. Thereafter, such formulations shall be regulated as non-scheduled formulations under paragraph 20.
Average price to retailer under paragraphs 4, 5 and 6 shall be calculated on dosage basis, and the ceiling price or retail price of a pack shall be determined by multiplying such unit price by the number or quantity contained in the pack. Where unit dosage is not specified in the First Schedule, the lowest pack size under the Drugs and Cosmetics Act, 1940 shall be treated as unit dosage for price calculation, including for paragraph 5 cases. For injections, inhalations, or formulations without specified dosage form or strength in Schedule I, the Government may notify separate ceiling or retail prices based on therapeutic rationale and pack parameters. The Government shall constitute a Committee of Experts within 15 days of the Order to recommend such separate price fixation.
A manufacturer, launching a scheduled formulation, shall be free to fix the price of the scheduled formulation equal Price list; to or below the ceiling price fixed for that schedule formulation by the Government. Where an existing brand is re-launched by another manufacturer the provisions of paragraph 13 shall be applicable.
Existing manufacturers selling scheduled formulations above the notified ceiling price plus applicable local taxes shall reduce such prices to not exceed that limit. For formulations already in the market, MRP shall be aligned within 45 days from the ceiling price notification.
All the existing manufactures of scheduled formulations, selling the branded or generic or both the versions of scheduled formulations at a price lower than the ceiling price (plus local taxes as applicable) so fixed and notified by the Government shall maintain their existing maximum retail price.
Annual increase in maximum retail price may be carried out as per the increase in the wholesale price index with respect to previous year as per the provision of sub-paragraphs (2) and (3) of paragraph 16.
Provided that in case of decline in wholesale price index, a corresponding reduction in the prices shall be made as per the provision of sub-paragraph (4) of paragraph 16.
Govt shall fix and notify ceiling prices of scheduled formulations in accordance with the provisions of the paragraphs 4 and 6, as the case may be & no manufacturer shall sell scheduled formulations at a price higher than ceiling price (plus local taxes as applicable) so fixed and notified by Govt.
Where any manufacturer sells a scheduled formulation at a price higher than the ceiling price (plus local taxes as applicable) fixed and notified by the Government, such manufacturers shall be liable to deposit the overcharged amount along with interest thereon from the date of such overcharging.
Where an existing manufacturer of a drug with dosages and strengths as specified in National List of Essential Medicines launches a new drug, such existing manufacturers shall apply for prior price approval of such new drug from the Government in Form-I specified under Schedule-II of this Order.
Manufacturers may revise the MRP of scheduled formulations once annually in April based on the previous calendar year’s wholesale price index without prior Government approval, and details of such revision shall be submitted to the Government in Form II, electronically or physically, within 15 days of revision.
On Wholesale Price Index decline, MRP must be reduced accordingly. For scheduled formulations already in the market, manufacturers must revise MRP within 45 days of notification of revised ceiling price to not exceed the new ceiling price (plus applicable taxes).
No manufacturers to increase MRP of drugs by more than 10% of MRP during the preceding 12 months. If the increase exceeds 10%, it must be reduced to the 10% limit and maintained at that level for the next 12 months.
The Government shall monitor the maximum retail prices (MRP) of all the drugs, including the non scheduled formulations and ensure that no manufacturer increases the maximum retail price of a drug more than ten percent of maximum retail price during preceding twelve months and where the increase is beyond ten percent of maximum retail price, it shall reduce the same to the level of ten percent of maximum retail price for next twelve months.
The manufacturer of scheduled formulations and the active pharmaceutical ingredients contained in the scheduled formulation shall furnish the information as stated in Form-III of schedule-II of DPCO 2013 quarterly.
Manufacturers planning to discontinue a scheduled formulation must give a public notice and inform the Government in Form-IV of Schedule II of DPCO 2013 at least six months before the intended discontinuation date.
Manufacturers shall reduce the MRP of scheduled formulations exceeding the ceiling price plus applicable local taxes to the latter. For formulations already produced or in the market, MRP shall be aligned within 45 days from the date of ceiling price notification.
For all the scheduled formulations having maximum retail price (MRP) higher than ceiling price (plus local taxes as applicable), the manufactures shall revise the maximum retail price (MRP) not exceeding the ceiling price (plus local taxes as applicable).
Further that in case of scheduled formulations produced or available in the market before the date of notification of ceiling price, the manufacturers shall ensure within a period of 45 days of the date of the notification that the maximum retail price of such scheduled formulation does not exceed the ceiling price (plus local taxes as applicable).
Every manufacturer of a schedule formulation intended for sale shall display in indelible print mark, on the label of container of the formulation and the minimum pack thereof offered for retail sale, the maximum retail price of that formulation based on the ceiling price notified in the Official Gazette or ordered by the Government in this behalf with the words “Maximum Retail Price” preceding it and the words ‘inclusive of all taxes’ succeeding it.
Every manufacturer of Scheduled/Non scheduled formulations shall issue price list and, if required, supplementary price list in Form V to dealers, State Drugs Controllers and the Government, indicating details of price fixation or revision as per notified order or Gazette. Every retailer and dealer shall display the price list and the supplementary price list, if any, as furnished by the manufacturer, on a conspicuous part of the premises where he carries on business in a manner so as to be easily accessible to any person wishing to consult the same.
Every manufacturer of scheduled/non-Scheduled formulations incl. NPPA regulated medical devices shall issue price list & if required, supplementary price list in Form VI to dealers, State Drugs Controllers, Government, indicating details of price fixation or revision as per notified order or Gazette Every retailer and dealer shall display the price list and the supplementary price list, if any, as furnished by the manufacturer, on a conspicuous part of the premises where he carries on business in a manner so as to be easily accessible to any person wishing to consult the same.
Every manufacturer of a non-Scheduled formulation intended for sale shall display in indelible print mark, on the label of container of the formulation and the minimum pack thereof offered for retail sale, the maximum retail price of that formulation with the words “Maximum Retail Price” preceding it and the words ‘inclusive of all taxes’ succeeding it.
No person shall sell any formulation to any consumer at a price exceeding the price specified in the current price list or price indicated on the label of the container or pack thereof, whichever is less.
No dealer shall sell loose quantity of any formulation at a price which exceeds the prorata price of the formulation.
Subject to the provisions of the Drug and CosmeticsAct, 1940 (23 of 1940) and the rules made thereunder,-
(a) no manufacturer or distributor shall withhold from sale or refuse to sell to a dealer any drug without good and sufficient reasons;
(b) no dealer shall with hold from sale or refuse to sell any drug available with him to a customer intending to purchase such drug.
Every manufacturer shall maintain records relating to the sales of individual active pharmaceutical ingredients or bulk drugs manufactured or imported and marketed by him, as the case may be, and sales of formulations units and packs and also such records as may be directed from time to time by Govt.
Penalty and Punishment
Para 14(2) – Drugs (Prices Control) Order, 2013
If a manufacturer sells a scheduled formulation above the notified ceiling price (plus applicable taxes), the overcharged amount along with applicable interest must be deposited with the Government from the date of overcharging.
Section 7 – Essential Commodities Act, 1955 (Penalties)
Contravention of orders issued under Section 3 attracts imprisonment (minimum 3 months up to 7 years) along with fine; additionally, the concerned goods, related packaging, and any vehicle or conveyance used may be forfeited to the Government.
Para 15(5) & 15(6) – Drugs (Prices Control) Order, 2013
Failure by an existing manufacturer to obtain prior price approval (Form-I) or selling a new drug above the Government-fixed retail price (plus applicable taxes) results in liability to deposit the overcharged amount along with interest from the date of launch/overcharge, in addition to applicable penalties.
Para 16(3) & 16(5) – Drugs (Prices Control) Order, 2013
Non-submission of required information is treated as non-revision or non-reduction of MRP, and the manufacturer becomes liable to deposit the excess amount charged over the applicable MRP (pre-revised or WPI-adjusted), along with interest from the date of overcharging.
Para 20(2) – Drugs (Prices Control) Order, 2013
If a manufacturer increases the price in violation of prescribed limits, the overcharged amount along with applicable interest from the date of such increase must be deposited, in addition to applicable penalties.
Para 23 – Drugs (Prices Control) Order, 2013
The Government may, by notice, require manufacturers, importers, or distributors to deposit the amount accrued from charging prices higher than those fixed or notified under the Drugs (Prices Control) Order, 1987 and 1995.
Disclaimer: The information contained in this Article is intended solely for personal non-commercial use of the user who accepts full responsibility of its use. The information in the article is general in nature and should not be considered to be legal, tax, accounting, consulting or any other professional advice. We make no representation or warranty of any kind, express or implied regarding the accuracy, adequacy, reliability or completeness of any information on our page/article.