
Introduction
Form 25A is the licence to manufacture drugs other than specified in Sch X, C and C(1) for the purpose of sale or distribution in India, issued under the Drugs and Cosmetics Rules, 1945.
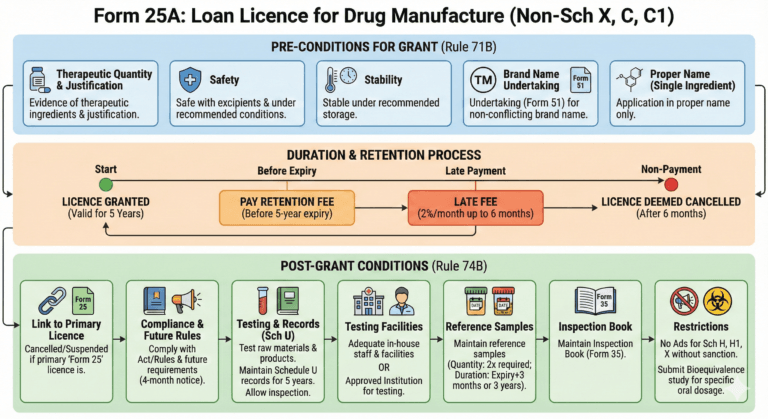
Conditions for the grant of a licence in Form 25A (Rule 71B)
Before a licence in Form 25A is granted, the applicant shall, while applying for a licence to manufacture drugs, furnish to the licensing authority evidence and date justifying that the drugs—
Provided that the application for grant of a licence for a drug formulation containing single active ingredient shall be made only in proper name.
Duration of loan licence
(1) A licence issued in Form 25A shall remain valid if licencee deposits a licence retention fee referred to in sub-rule (2) before the expiry of a period of every succeeding five years from the date of its issue, unless, it is suspended or cancelled by the licensing authority
(2) The licence retention fee referred to in sub-rule (1) shall be equivalent to the respective fee required for the grant of such licence excluding inspection fee paid for grant of licence.
(3) If the licencee fails to pay licence retention fee on or before the due date as referred to in sub-rule (1), he shall be liable to pay licence retention fee along with a late fee calculated at the rate of two per cent, of the licence fee for every month or part there of up to six months, and in the event of non-payment of such fee, the licence shall be deemed to have been cancelled.
74B. Conditions of licence in Form 25A
Disclaimer: The information contained in this Article is intended solely for personal non-commercial use of the user who accepts full responsibility of its use. The information in the article is general in nature and should not be considered to be legal, tax, accounting, consulting or any other professional advice. We make no representation or warranty of any kind, express or implied regarding the accuracy, adequacy, reliability or completeness of any information on our page/article.