
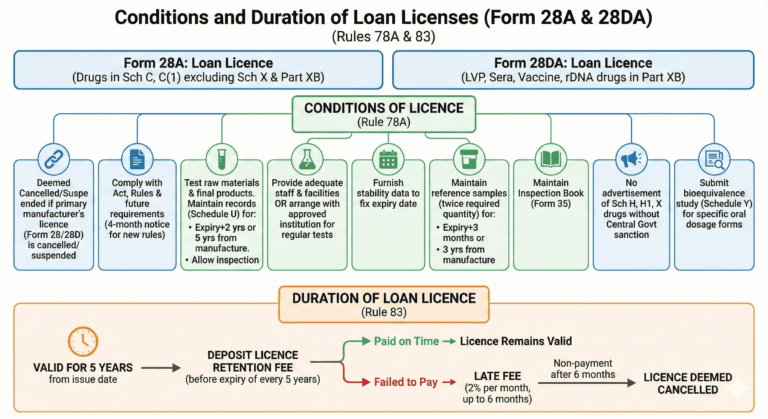
Form 28A: Loan Licence to manufacture drugs specified in Sch C, C(1) excluding Sch X and Part XB
Loan licences for drugs under Sch C & C(1), excluding Sch X, LVPs, Sera, and r-DNA drugs, must be in Form 28A. Applicants must justify safety, efficacy, stability, and therapeutic purpose. In case of brand name, applicant would require Form 51 to be submitted to authority.
Form 28DA: Loan Licence to manufacture LVP, Sera and Vaccine and rDNA drugs specified in Part XB
Loan Licence to manufacture LVP, Sera and Vaccine and rDNA drugs specified in Part XB must be in Form 28A. Applicants must justify safety, efficacy, stability, and therapeutic purpose. In case of brand name, applicant would require Form 51 to be submitted to authority.
Conditions of licence in Form 28A or Form 28DA (Rule 78A)
(1) The licence in Form 28A or Form 28DA shall be deemed to be cancelled or suspended, if the licence owned by the licensee in Form 28 or Form 28D whose manufacturing facilities have been availed of by the licensee is cancelled or suspended, as the case may be, under these rules.
(2) The licensee shall comply with the provisions of the Act, and of these rules and with such further requirements if any, as may be specified in any rules subsequently made under Chapter IV of the Act, provided that where such further requirements are specified in the rules, those would come into force four months after publication in the Official Gazette.
(3) The licensee shall test each batch or lot of the raw material used by him for the manufacture of his products and also each batch of the final product and shall maintain records or registers showing the particulars in respect of such tests as specified in Schedule U. Records or registers shall be retained, in the case of a substance for which a potency date is fixed, for a period of two years from the expiry of such date and in the case of other substances, for a period of five years from the date of manufacture. The licensee shall allow an Inspector to inspect all registers and records maintained under these rules and shall supply to the Inspector such information as he may require for the purpose of ascertaining whether the provisions of the Act and these rules have been observed.
(4) The licensee shall either (i) provide and maintain to the satisfaction of the licensing authority adequate staff and adequate laboratory facilities for carrying out tests of the strength, quality and purity of the substances manufactured by him. or (ii) make arrangements with some institution approved by the licensing authority for such tests to be regularly carried out on his behalf by the institution.
(5) The licensee shall furnish to the licensing authority, if required to do so, data on the stability of drugs which are likely to deteriorate for fixing the date of expiry which would be printed on the labels of such drugs on the basis of the date so furnished.
(6) The licensee shall maintain reference samples from each batch of the drugs manufactured by him in a quantity which is at least twice the quantity of the drug required to conduct all the tests performed on the batch. In case of drugs bearing an expiry date on the labels, the reference samples shall be maintained for a period of three months beyond the date of expiry of potency. In case of drugs where no date of expiry of potency is specified on the label, the reference samples shall be maintained for a period of three years from the date of manufacture.
(7) The licensee shall maintain an Inspection Book in Form 35 to enable an Inspector to record his impressions and the defects noticed.
(8) No advertisement of the drugs specified in Schedule H, Schedule HI or Schedule X shall be made except with the previous sanction of the Central Government.
(9) the applicant shall submit the result of bioequivalence study referred to in Schedule Y, along with the application for grant of a licence of oral dosage form of drugs specified under category II and category TV of the biopharmaceutical classification system.
Duration of loan licence (Rule 83)
(1) A loan licence issued in Form 28A and Form 28DA shall remain valid, if the licencee deposits a licence retention fee referred to in sub-rule (2) before the expiry of period of every succeeding five years from the date of its issue, unless it is suspended or cancelled by the licensing authority.
(2) The licence retention fee referred to in sub-rule (1) shall be equivalent to the respective fee required for the grant of such licence excluding inspection fee paid for grant of licence.
(3) If the licencee fails to pay licence retention fee on or before the due date as referred to in sub-rule (1), he shall be liable to pay licence retention fee along with a late fee calculated at the rate of two per cent, of the licence fee for every month or part thereof up to six months, and in the event of non-payment of such fee, the licence shall be deemed to have been cancelled.
Disclaimer: The information contained in this Article is intended solely for personal non-commercial use of the user who accepts full responsibility of its use. The information in the article is general in nature and should not be considered to be legal, tax, accounting, consulting or any other professional advice. We make no representation or warranty of any kind, express or implied regarding the accuracy, adequacy, reliability or completeness of any information on our page/article.