
Approval for Clinical Trials of New/Investigational New Drugs by Ethics Committee & CLA (Rule 19)
(1) No person or institution or organisation shall conduct clinical trial of a new drug or investigational new drug,—
(i) except in accordance with the permission granted by the Central Licencing Authority; and
(ii) without the protocol thereof having been approved by the Ethics Committee for clinical trial registered in accordance with the provisions of rule 8.
(2) Every person associated with the conduct of clinical trial of a new drug or investigational new drug shall follow the general principles and practices as specified in the First Schedule.
(3) No person or institution or organisation shall conduct clinical trial of a new drug or investigational new drug except in accordance with the procedure prescribed under the provisions of the Act and these rules.
Form CT-06: Permission to Conduct Clinical Trial of New Drug or Investigational New Drug (Rule 21, 22(1), (2), 23, 24)
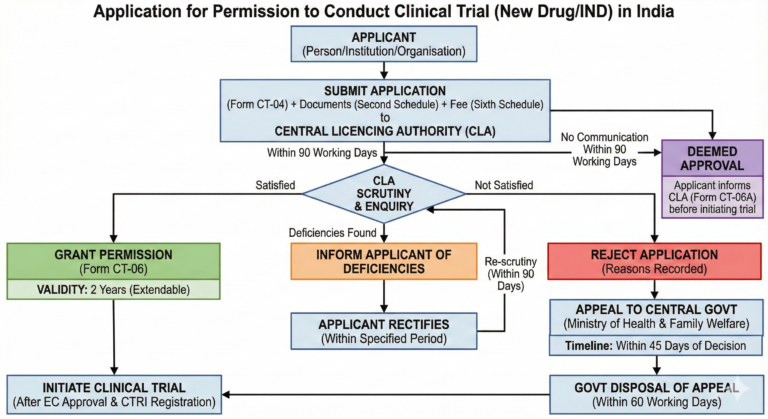
Rule 21:-
To conduct a clinical trial of a new or investigational drug, an application in Form CT-04 must be submitted to the Central Licensing Authority, along with required documents and prescribed fee. An institution or organisation funded or owned, wholly or partially by the Central Government or by a State Government are exempt from paying the fee.
Rule 23:
(1) Notwithstanding these rules, if a complete application is submitted under Rule 21 to conduct a clinical trial of a new or investigational drug that meets the following conditions:-
(i) drug is discovered in India;
(ii) If the drug’s research/development/manufacturing/and marketing are to be done in India, the Central Licensing Authority shall process the application by granting permission, rejecting it, or requesting corrections within 30 working days from receiving the application, as specified in Rule 22.
If the Central Licensing Authority does not communicate with the applicant within the specified period, permission to conduct the clinical trial is deemed granted & the applicant is authorized to proceed as per the rules. This permission is legally valid for all purposes.
(2) The applicant with deemed approval under sub-rule (1) must inform the CLA in Form CT-4A before starting the clinical trial. The Authority will record the Form CT-4A, which will serve as automatic approval
Rule 24:
Notwithstanding anything contained in these rules, where any person or institution or organisation makes an application under rule 21 to conduct clinical trial of a new drug which is already approved and marketed in a country, as specified under rule 101, the application, shall be disposed of by way of grant of permission or rejection or processed by way of communication to rectify any deficiency, as the case may be, as specified in rule 22, by the Central Licencing Authority within a period of ninety working days from the date of the receipt of the application by the said Authority.Provided that, where no communication has been received from the Central Licensing Authority within the said period of ninety working days, the permission to conduct clinical trial shall be deemed to have been granted by the
Central Licensing Authority and such permission shall be deemed to be legally valid for all purposes and the applicant shall be authorised to initiate clinical trial in accordance with these rules.Provided further that the applicant who has got deemed approval under this rule shall before initiating the clinical trial, inform the Central Licensing Authority in Form CT-06A and the Central Licensing Authority shall on the
basis of the said information, take on record the Form CT-06A which shall become part of the official record and shall be called deemed approval of the Central Licensing Authority
Form CT-06A: Information to initiate Clinical Trial of New Drug or Investigational New Drug (Rule 22 (2A), 24)
.The applicant who has got deemed approval under the proviso to sub-rule(2) shall, before initiating the clinical trial, inform the Central Licensing Authority in Form CT-06A and the Central Licensing Authority shall on the basis of the said information, take on record the Form CT-06A which shall
become part of the official record and shall be called deemed approval of the Central Licensing Authority
Rule 24 – If a Rule 21 application for a clinical trial of a drug approved in a Rule 101 country isn’t processed within 90 working days, approval is deemed granted. The applicant must submit Form CT-06A, which the CLA will record as approval.
Six-Monthly Clinical Trial Status Report Submission to CLA (Rule 25 (viii))
Six monthly status report of each clinical trial, as to whether it is ongoing, completed or terminated, shall be submitted to the Central Licencing Authority electronically in the SUGAM portal.
Intimation of Termination of Clinical Trial to the CLA (Rule 25 (ix))
In case of termination of any clinical trial the detailed reasons for such termination shall be communicated to the Central Licencing Authority within thirty working days of such termination;
Post-trial access of investigational new drug or new drug (Rule 27)
Where any investigator of a clinical trial of investigational new drug or new drug has recommended post-trial access of the said drug after completion of clinical trial to any trial subject and the same has been approved by the Ethics Committee for clinical trial, the post-trial access shall be provided by the sponsor of such clinical trial to the trial subject free of cost:-
(i) if the clinical trial is being conducted for an indication for which no alternative therapy is available and the investigational new drug or new drug has been found to be beneficial to the trial subject by the investigator; and
(ii) the trial subject or legal heir of such subject, as the case may be, has consented in writing to use post-trial investigational new drug or new drug and the investigator has certified and the trial subject or his legal heir, as the case may be, has declared in writing that the sponsor shall have no liability for post trial use of investigational new drug or new drug.
Appeal against Suspension or cancellation of permission to conduct clinical trial (Rule 30 (2))
(1) Where any person or institution or organisation to whom permission has been granted under rule 22 in Form CT-06 or rule 23 in Form CT-4A fails to comply with any provision of the Act and these rules, the Central Licencing Authority may, after giving an opportunity to show cause and after affording an opportunity of being heard, by an order in writing, take one or more of the following actions, namely:—
(i) issue warning in writing describing the deficiency or defect observed during inspection or otherwise, which may affect adversely the right, or well- being of a trial subject or the validity of clinical trial conducted;
(ii) reject the results of clinical trial;
(iii) suspend for such period as considered appropriate or cancel the permission granted under rule 22 in Form CT-06 or rule 23 in Form CT-4A;
(iv) debar the investigator or the sponsor including his representatives to conduct any clinical trial in future for such period as considered appropriate by the Central Licencing Authority.
(2) Where a person or an institution or an organisation to whom permission has been granted under rule 22 in Form CT- 06 or rule 23 in Form CT-4A or the sponsor is aggrieved by the order of the Central Licencing Authority, the person or the institution or the organisation may, within a period of sixty working days of the receipt of the order, make an appeal to the Central Government and that Government may, after such enquiry, as deemed necessary, and after affording an opportunity of being heard, pass such order in relation thereto as may be considered appropriate in the facts and circumstances of the case.
Disclaimer: The information contained in this Article is intended solely for personal non-commercial use of the user who accepts full responsibility of its use. The information in the article is general in nature and should not be considered to be legal, tax, accounting, consulting or any other professional advice. We make no representation or warranty of any kind, express or implied regarding the accuracy, adequacy, reliability or completeness of any information on our page/article.