
Background
Under the Drugs and Cosmetics Act, 1940, registration under Form 41 is a mandatory requirement for establishments involved in manufacturing, importing, or wholesaling drugs. This registration once issued serves as confirmation that the premises meet the prescribed quality, safety, and Good Manufacturing Practices (GMP) standards. Failure to secure this registration and continue operations as required is treated as a serious offense, since it undermines public health and safety by potentially allowing the manufacture or distribution of substandard or unsafe drugs.
In legal purview, “Registration Certificate” means a certificate issued under rule 27A by the licensing authority in Form 41 for registration of the premises and the drugs manufactured by the manufacturer meant for import into and use in India.
Form and manner of application for Registration Certificate (Rule 24A)
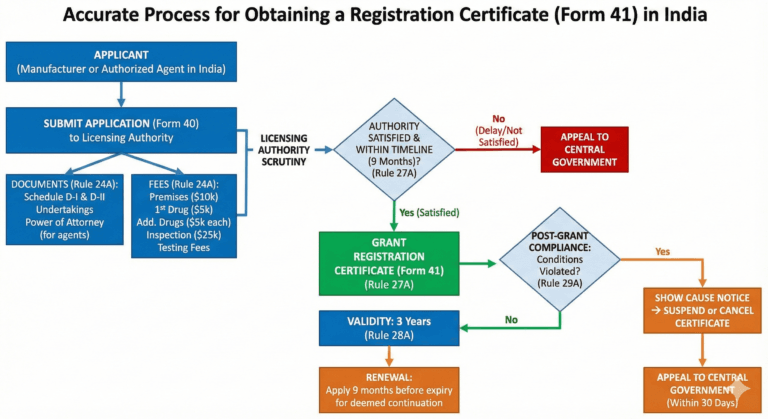
(1) An application for issue of a Registration Certificate shall be made to the licensing authority in Form 40, either by the manufacturer himself, having a valid whole sale licence for sale or distribution of drugs under these rules, or by his authorised agent in India, either having a valid licence under the rules to manufacture for sale of a drug or having a valid whole sale licence for sale or distribution of drugs under these rules, and shall be accompanied by the fee specified in sub-rule (3) and the informations and undertakings specified in Schedules D-I and D-II duly signed by or on behalf of the manufacturer.
(2) The authorisation by a manufacturer to bis agent in India shall be documented by a power of attorney executed and authenticated either in India before a First-Class Magistrate, or in the country of origin before such an equivalent authority, the certificate of which is attested by the Indian Embassy of the said country, and the original of the same shall be furnished alongwith the application for Registration Certificate.
(3) (i) A fee of ten thousand US dollars or its equivalent in Indian rupees shall be paid alongwith the application in Form 40 as registration fee for his premises meant for manufacturing of drugs intended for import into and use in India.
(ii) A fee of five thousand US dollars or its equivalent in Indian rupees] shall be paid alongwith the application in Form 40 for the registration of a single drug meant for import into and use in India and an additional fee at the rate of five thousand US dollars for each additional drug:
Provided that in the case of any subsequent application for registration of additional drugs by the same manufacturer, the fee to accompany shall be five thousand US dollars or its equivalent in Indian rupees for each drug.
(4) The fees shall be paid through a Challan in the Bank of Baroda, Kasturba Gandhi Marg, New Delhi-110 001 or any other branch or branches of Bank of Baroda, or any other bank, as notified, from time to time, by the Central Government, to be credited under the Head of Account “0210-Medical and Public Health, 04-Public Health, 104-Fees and Fines”:
Provided that in the case of any direct payment of fees by a manufacturer in the country of origin, the fees shall be paid through Electronic Clearance System (ECS) from any bank in the country of origin to the Bank of Baroda, Kasturba Gandhi Marg, New Delhi, through the Electronic Code of the bank in the Head of Account “0210-Medical and Public Health, 04-Public Health, 104- Fee and Fines”, and the original receipt of the said transfer shall be treated as an equivalent to the bank challan, subject to the approval by the Bank of Baroda that they have received the payment.
(5) The applicant shall be liable for the payment of a fee of twenty-five thousand US dollars or its equivalent in Indian rupees for expenditure as may be required for inspection or visit of the manufacturing premises of drugs, by the be required for inspection or visit of the manufacturing premises of drugs, by the licensing authority or by any other persons to whom powers have been delegated in this behalf by the licensing authority under rule 22:
(6) The applicant shall be liable for the payment of testing fees directly to a testing laboratory approved by the Central Government in India or abroad, as may be required for examination, tests and analysis of drug.
(7) A fee of one thousand eight hundred US dollars or its equivalent in Indian rupees shall be paid for making amendment in the registration certificate or for a duplicate copy of the Registration Certificate, if the original is defaced, damaged or lost.
(8) No Registration Certificate shall be required under these rules in respect of an inactive bulk substance to be used for a drug formulation, with or without pharmacopeal conformity.
Registration Certificate for import of drugs manufactured by one manufacturer (Rule 25B)
(1) A single application may be made, and a single Registration Certificate in Form 41 may be issued in respect of the import of more than one drug or class of drugs, manufactured by the same manufacturer:
Provided that the drug or classes of drugs, are manufactured at one factory or more than one factory functioning conjointly as a single manufacturing unit:
Provided further that if a single manufacturer has two or more factories situated in different places manufacturing the same or different drugs, separate Registration Certificates shall be required in respect of the drugs manufactured by each such factory.
Grant of Registration Certificates (Rule 27A)
(1) On receipt for an application for Registration Certificate in the Form and manner specified in rule 24A, the licensing authority shall, on being satisfied, that, if granted, the conditions of the Registration Certificate will be observed, issue a Registration Certificate in Form 41:
Provided further that if the application is complete in all respects and informations specified in Schedules DI and DII are in order, the licensing authority shall, within nine months from the date of receipt of an application, issue such Registration Certificate, and in exceptional circumstances and for reasons to be recorded in writing, the Registration Certificate may be issued within such extended period, not exceeding three months as the licensing authority, may deem fit.
(2) If the applicant does not receive the Registration Certificate within the period as specified in proviso to sub-rule (1), he may appeal to the Central Government and the Central Government may after such enquiry into the matter, as it considers necessary, may pass such orders in relation thereto as it thinks fit.
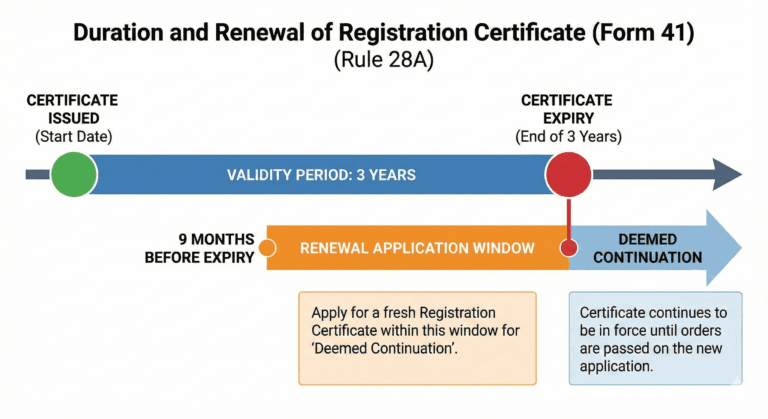
Duration of Registration Certificate (Rule 28A)
A Registration Certificate, unless, it is sooner suspended or cancelled, shall be valid for a period of three years from the date of its issue:
Provided that if the application for a fresh Registration Certificate is made nine months before the expiry of the existing certificate, the current Registration Certificate shall be deemed to continue in force until orders are passed on the application.
Suspension and cancellation of Registration Certificate (Rule 29A)
If the manufacturer fails to comply with any of the conditions of the Registration Certificate, the licensing authority may after giving him an opportunity to show cause why such an order should not be passed, by an order in writing stating the reasons therefor, suspend or cancel the Registration Certificate for such period as it thinks fit either wholly or in respect of some of the substances to which it relates:
Provided that a person, who is aggrieved by the order passed by the licensing authority under this rule may, within thirty days of the receipt of the order, appeal to the Central Government, and the Central Government may, after such enquiry into the matter as it considers necessary and after giving the appellant an opportunity for representing his views in the matter, pass such orders in relation thereto as it thinks fit.
Disclaimer: The information contained in this Article is intended solely for personal non-commercial use of the user who accepts full responsibility of its use. The information in the article is general in nature and should not be considered to be legal, tax, accounting, consulting or any other professional advice. We make no representation or warranty of any kind, express or implied regarding the accuracy, adequacy, reliability or completeness of any information on our page/article.