
(Applicable to licences in Forms 20, 20A, 20B, 20F, 20G, 21 and 21B under the Drugs Rules, 1945)
The licences listed above shall be governed by the conditions stated in the respective licence forms and the following general conditions as per Rule 65:
Compounding and Supply under Pharmacist Supervision
Record of Supply on Prescriptions
When supplying a drug (other than Schedule X) on the prescription of a Registered Medical Practitioner, the following must be recorded at the time of supply in a Prescription Register:
Permitted alternatives:
Exemptions from detailed entry:
Schedule H1
Declaration of method
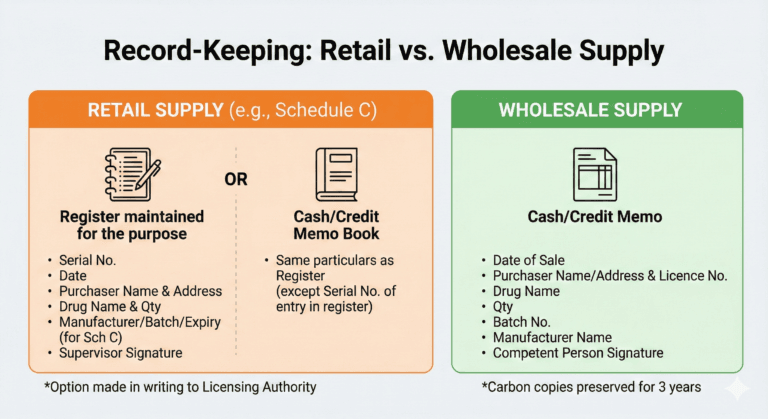
Supply by Retail Without Prescription — Schedule C Drugs
Retail supply without prescription must be recorded either:
Licensing Authority may mandate register-only maintenance if memo copies are not legible.
Carbon copies must be retained.
Records of Purchases – Retail Sales
Licensees must maintain purchase records showing:
Purchase bills / memos must be serially numbered and filed chronologically.
Supply by Wholesale
Wholesale dealing must be accompanied by a cash/credit memo showing:
Carbon copies must be retained for 3 years.
Wholesale purchase records must meet the same requirements as retail purchase records.
Inspection of Records
All required registers and records must be:
Record Retention
Unless otherwise stated,
Duplication Avoidance
Where required particulars are already recorded in another register maintained under any other applicable law, separate entries in a drug register are not necessary.
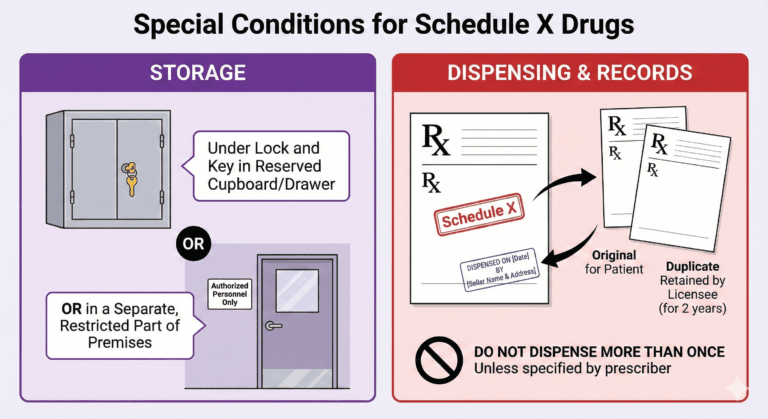
Special Controls — Schedules H, H1, and X
Dispensing requirements:
Schedule X storage requirements:
Disclaimer: The information contained in this Article is intended solely for personal non-commercial use of the user who accepts full responsibility of its use. The information in the article is general in nature and should not be considered to be legal, tax, accounting, consulting or any other professional advice. We make no representation or warranty of any kind, express or implied regarding the accuracy, adequacy, reliability or completeness of any information on our page/article.