
Introduction
The import of drugs in India is strictly regulated to ensure quality, safety, and efficacy. Central to this regulatory framework is the requirement for an import license, which must be obtained prior to bringing drugs into the country. Applicants use Form 10 (or Form 10A for drugs specified under Schedule X) to secure such a license. Both forms are issued by the Central Drugs Standard Control Organization (CDSCO) and serve to validate that the importer meets all regulatory requirements. The following procedure note details the step-by-step process for obtaining an import license using these forms.
Conditions to be satisfied before a licence in Form 10 or Form 10A is granted (Rule 25A)
(1) A licence in Form 10 or in Form 10A shall be granted by the licensing authority having regard to—
(i) the premises, where the imported substances will be stocked are equipped with proper storage accommodation for preserving the properties of the drugs to which the licence applies; and
(ii) the occupation, trade or business ordinarily carried out by the applicant:
Provided that the licensing authority may refuse to grant a licence in Form 10A in respect of any applicant where he is satisfied—
(a) that the applicant has not complied with the provisions of the Act or these rules, or
(b) that by reasons of—
(i) his conviction under the Act or these rules or the Narcotic Drugs and Psychotropic Substances Act, 1985 (61 of 1985) or the rules made thereunder;
(ii) previous suspension or cancellation of the licence granted to him, he is not a fit person to whom licence shall be granted.
(2) Any person who is aggrieved by the order passed by the licensing authority under this rule may, within thirty days of the receipt of the order, appeal to the Central Government and the Central Government may after such enquiry into the matter as it considers necessary and after giving the appellant an opportunity for making a representation in the matter, make such orders in relation thereto as it thinks fit.
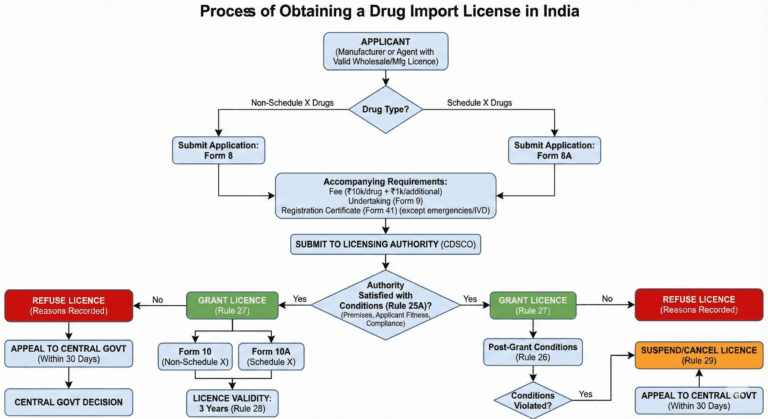
Form and manner of application for import licence (Rule 24)
(1) An application for an import licence shall be made to the licensing authority in
Form 8 for drugs excluding those specified in Schedule X, and in
Form 8A for drugs specified in Schedule X,
either by the manufacturer himself having a valid wholesale licence for sale or distribution of drugs under these rules, or by the manufacturer’s agent in India either having a valid licence under the rules to manufacture for sale of a drug or having a valid wholesale licence for sale or distribution of drugs under these rules, and shall be accompanied by a licence fee of ten thousand rupees for a single drug and an additional fee at the rate of one thousand rupees for each additional drug and by an undertaking in Form 9 duly signed by or on behalf of the manufacturer:
Provided that in the case of any subsequent application made by the same importer for import licence for drugs manufactured by the same manufacturer, the fee to accompany each such application shall be one thousand rupees for each drug.
(2) Any application for import licence in Form 8 or Form 8A, as the case may be, shall be accompanied by a copy of Registration Certificate issued in Form 41 under rule 27A:
Provided that in case of emergencies the licensing authority may, with the approval of the Central Government, issue an import licence in Form 10 or 10A, as the case may be, without the issuance of Registration Certificate under rule 27A, for reasons to be recorded in writing:
Provided further that Registration Certificate shall not be required to be accompanied with an application for an import licence under the rules for the import of in-vitro diagnostic kits and regents, except for the diagnostic kits notified from time to time under sub-clause (iv) of clause (b) of section 3.
(3) A fee of one thousand five hundred rupees shall be paid for making amendment in the licence
Licences for import of drugs manufactured by one manufacturer (Rule 25)
(1) A single application may be made, and a single licence may be issued, in respect of the import of more than one drug or class of drugs manufactured by the same manufacturer:
Provided that the drugs or classes of drugs are manufactured at one factory or more than one factory functioning conjointly as a single manufacturing unit:
Provided further that if a single manufacturer has two or more factories situated in different places manufacturing the same or different drugs a separate licence shall be required in respect of the drugs manufactured by each such factory.
Conditions of import licence (Rule 26)
An import licence shall be subject to the following conditions:—
(i) the manufacturer shall at all times observe the undertaking given by him or on his behalf in Form 9;
(ii) the license shall allow any Inspector authorized by the licensing authority in that behalf to enter with or without notice any premises where the imported substance is stocked to inspect the means, if any, employed for testing the substance and to take samples;
(iii) the licensee shall on request furnish to the licensing authority from every batch of each substance or from such batch or batches at the licensing authority may from time to time specify a sample of such amount as the licensing authority may consider adequate for any examination required to be made, and the licensee shall, if so required, furnish full protocols of the tests, if any, which have been applied;
(iv) if the licensing authority so directs the licensee shall not sell or offer for sale any batch in respect of which a sample is or protocols are furnished under the last preceding sub-rule until a certificate authorizing the sale of the batch has been issued to him by or on behalf of the licensing authority;
(v) the licensee shall, on being informed by the licensing authority that any part of any batch of the substance has been found by the licensing authority not to conform with the standards of strength, quality and purity prescribed by Chapter III of the Act, or the Rules thereunder and on being directed so to do, withdraw the remainder of that batch from sale and, so far as may in the particular circumstances of the case be practicable, recall the issues already made from that batch;
(vi) the licensee shall maintain a record of all sales by him of substances for the import of which a license is required, showing particulars of the substance and of the person to whom sold and such further particulars, if any, as the licensing authority may specify and such record shall be open to the inspection of any Inspector authorized in that behalf by the licensing authority: Provided that in respect of the sale or distribution of drugs specified in Schedule X, the licensee shall maintain a separate record or register showing the following particulars, namely:—
(vii) the licensee shall comply with such further requirements, if any, applicable to the holders of import licences, as may be specified in any rules, subsequently made under Chapter III of the Act and of which the licensing authority has given to him not less than four months’ notice.
Grant of import licence (Rule 27)
On receipt of an application for an import licence in the Form and manner prescribed in rule 24, the licensing authority shall, on being satisfied that, if granted, the conditions of the licence will be observed, issue an import licence in Form 10 or Form 10A, as the case may be.
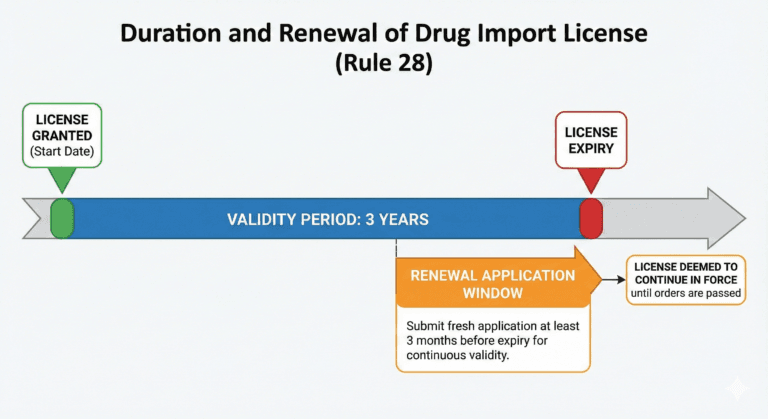
Duration of import licence (Rule 28)
A licence, unless, it is sooner suspended or cancelled, shall be valid for a period of three years from the date of its issue
Provided that if application for a fresh licence is made three months before the expiry of the existing licence the current licence shall be deemed to continue in force until orders are passed on the application.
Suspension and cancellation of import licence (Rule 29)
If the manufacturer or licensee fails to comply with any of the conditions of an import licence, the licensing authority may after giving the manufacturer or licensee an opportunity to show cause why such an order should not be passed, by an order in writing stating the reasons therefor, suspend or cancel it for such period as it thinks fit either wholly or in respect of some of the substances to which it relates:
Provided that a person who is aggrieved by the order passed by the licensing authority under this rule may, within thirty days of the receipt of the order, appeal to the Central Government, and the Central Government may, after such enquiry into the matter, as it considers necessary and after giving the said appellant an opportunity for representing his views, pass such orders in relation thereto as it thinks fit.
Disclaimer: The information contained in this Article is intended solely for personal non-commercial use of the user who accepts full responsibility of its use. The information in the article is general in nature and should not be considered to be legal, tax, accounting, consulting or any other professional advice. We make no representation or warranty of any kind, express or implied regarding the accuracy, adequacy, reliability or completeness of any information on our page/article.